Engineering healthy ageing with stem cells
How UQ researchers are engineering artificial human tissues to activate stem cells, regenerate tissue and fight ageing.

Artificially creating new tissue for the body has been an important focus for modern science.
Getting it right is critical, whether a person has lost tissue, like after a mastectomy, or it's been injured or simply gets old.
UQ’s Professor Justin Cooper-White has united with world experts from Stanford, Harvard and the University of Pennsylvania to create special materials that will near perfectly match the unique properties of our own body.
By embracing the surprising fact our body tissues are elastic and solid, but also work a bit like a liquid, he is working towards therapies that improve our ability to heal and could even reverse the effects of ageing.
In early 2000, an eminent surgeon came knocking at the door of the Department of Chemical and Biomolecular Engineering at The University of Melbourne.
He was looking for a tissue engineer to help him create biological materials for surgery.
“In the late nineties, tissue engineering was a fledgling field in Australia. Almost unknown. So, there were no tissue engineers in the department to be found,” recalls Professor Justin Cooper-White, Head of UQ's School of Chemical Engineering.
At the time, Professor Cooper-White was a young chemical engineer, working as a postdoctoral researcher at The University of Melbourne.
“I had done my PhD on the properties of a particular kind of degradable plastic,” Professor Cooper-White said.
“My head of department knew researchers in the United States were using this material for biomedical applications, so he suggested the surgeon talk to me.”
Professor Cooper-White’s research to date had nothing to do with biomedicine, but the universe had other plans.
“That started my collaboration with the wonderful microsurgeon, Professor Wayne Morrison, at St Vincent's Hospital in Melbourne,” he said.
“Together, we created new materials for breast augmentation in mastectomy victims.”
Rather than just dropping in a bag of silicone, as was standard practice at the time, the pair had a completely different approach in mind.
“We took materials normally used in degradable stitches and screws and created structures out of them that were like a foam, similar to sponges we wash up with.”
This material was surgically implanted to replace missing breast tissue.
“You create this big porous ‘scaffold’ that allows a person’s own cells to crawl into and colonise. Those cells can then start creating whole new tissues. The original purpose was just to create space for this to happen.”
These spaces were designed to mimic similar spaces found naturally inside our bodies.


“If you look at a thin slice of breast tissue or muscle under a microscope, you see a lot of cells," Professor Cooper-White explains.
“Around those cells are proteins made by those same cells. These proteins make a big framework called the ‘extracellular matrix’. Extracellular meaning ‘outside the cell’.
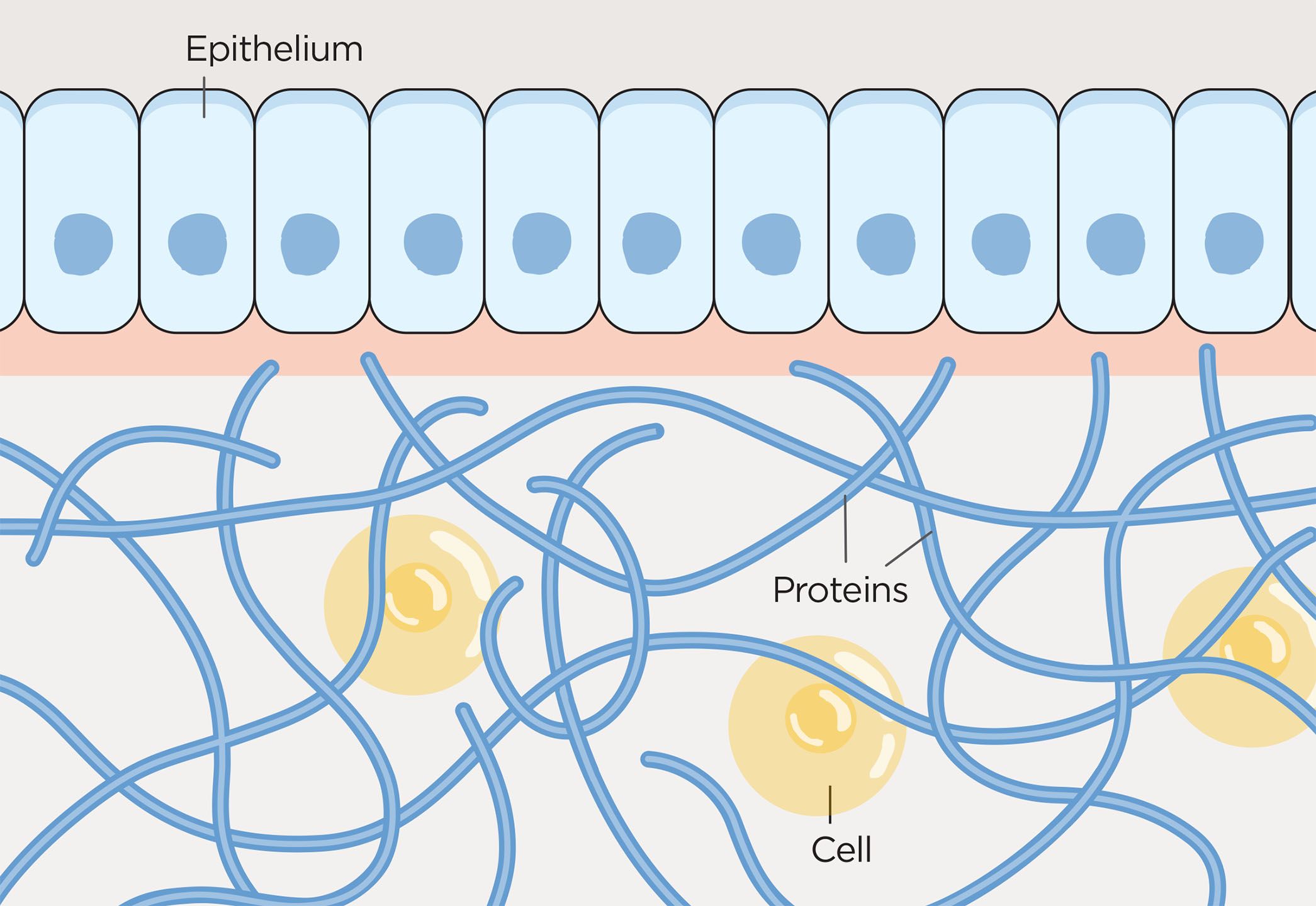
Cells (yellow circles) can be seen sitting in a web of extra-cellular matrix, formed of proteins (blue lines). The epithelium forms a barrier of cells between the body and outer world.
Cells (yellow circles) can be seen sitting in a web of extra-cellular matrix, formed of proteins (blue lines). The epithelium forms a barrier of cells between the body and outer world.
“It’s this matrix we are replacing in breast reconstruction. We create a home for when cells have lost their home.”
Their early work showed this process could also work with other kinds of tissue, like bone, cartilage, muscle, kidney and even brain tissue.
You simply make an artificial space, and wait for the cells to arrive.
If you build it, will they come?
While making incredible strides, Professor Cooper-White and his team began to realise there might be more to tissue engineering than just creating a space.
They saw some properties of the matrices they created had big effects on the cells that moved into them.
“If you think about a home, a home isn’t just about building the frame of the house,” Professor Cooper-White explains.
“You've got to fill it with nice furnishings and appropriate wallpaper. Maybe a painting or two.
“This helps somebody who lives inside the house feel comfortable. They associate with their environment. Our cells are exactly the same.
“We realised we didn't really understand how cells engaged with that synthetic environment we'd created for them.”
Evidence coming out of the US seemed to suggest how spongey or stiff a matrix was could change how the cells that populated the matrix behaved.
This was seen in the way stem cells, ‘blank slate’ cells that can turn into various other types of cells, changed their form when added to different bodily tissues.
“Even though these results were derived on a lab bench, not in living bodies, they made a lot of sense”, Professor Cooper-White said.
“Other researchers injected stem cells into the ‘soft’ tissues in mice, such as into skeletal and heart muscle tissues. Where stem cells were injected into healthy areas of tissue, muscle was formed.”
“However, where the muscle was scarred and stiff from damage, the stem cells were unable to turn into healthy muscle effectively. The stiff scar tissue prevents them from achieving their full potential, as it turns out.”
The world took this to mean that a matrix had to be spongey for its stem cell residents to turn into the right kind of spongey cells.
But Professor Cooper-White, now at UQ, still felt like a piece of the puzzle was missing.
Inside the matrix
Cooper-White saw these different tissues were indeed spongey or stiff, but they also contained different proteins – large molecules made up of one or more chains of amino acids.
To test whether proteins were important in influencing how cells behave, Professor Cooper-White and his team designed an experiment.
They made a spongey matrix for cells, but then added the proteins usually found in a stiff matrix. Conversely, they made stiff matrices filled with spongey proteins.
“We showed that if you put a protein that is not in bone into a bone matrix, the stem cells won’t turn into bone at all. They turn into the tissue cell type that the protein is most similar to in its make-up,” Professor Cooper-White explained.
“Just making these matrix spaces of a particular stiffness is not good enough. They have to be made of the right protein as well.”
Having discovered how we can teach stem cells to do what we want them to do, Professor Cooper-White wondered whether we could use them to repair stiff, ageing or diseased tissue back to its spongey, healthy state.




Reversing the creep of time
One of Professor Cooper-White’s most important discoveries was that cells in our tissues respond really well when the matrix they are in is matched closely to our natural body tissue state, in terms of how the matrix’s deals with force and motion.
Human tissue, rather surprisingly, turns out to be rather like silly putty. It bounces, because it has elasticity. But it also loses its shape over time because it is liquid-like, or ‘viscous’, in nature.

Professor Cooper-White says you can demonstrate this special property of our tissues, known as ‘viscoelasticity’, with a simple experiment.
“If you try to touch your toes, you will bend down and get held up. That's because the muscles in your legs are relaxed initially and then when you put load on them by trying to touch your toes, they get pulled tight. This is the muscle’s elastic response.
“But, if you keep holding there, your muscle then starts to extend. This ‘creep’ in your muscles is related to the viscous response – the ability of the muscle to change its length a little at a time, without breaking.”
Looking to the future, Professor Cooper-White and his lab are investigating how the viscoelasticity of our bodies changes over time.
“We age because our body gets less viscoelastic. This is something we are focusing on now. Maybe it’s just because I am getting older.”
As we age, daily ailments cause inflammation. Inflammation in one area is carried around the whole body by our blood vessels, causing the extracellular matrix of all our tissues to become stiff and corrupted, or ‘fibrotic’.
This also corrupts the stem cell ‘niches’, the homes of stem cells that live next to our blood vessels.
“We’re investigating what happens when those tissues become less viscoelastic, and how we might reverse that,” Professor Cooper-White said.
“The question we’re exploring is how can we change that fibrotic state of all our tissues to a younger, more viscoelastic state?”
One way Professor Cooper-White and his team are doing this is by creating tiny particles containing genetic material with parts of our genome that reduce in expression as we get older.

“We deliver these nanoparticles and they reset the cells so they start producing a more normal extracellular matrix. They no longer produce fibrotic tissue,” he said.
Another approach targets stem cells themselves.
“We are also trying to return the stem cell niches, and therefore basically our vascular system, back to a younger state. If we do that, the stem cells will be able to function better and we will have overall better healing.”
In a recent review paper, published in the prestigious journal Nature, Professor Cooper-White has come together with other top researchers in the field to explain the crucial, but under-appreciated, role of viscoelasticity in health, disease and ageing to the world.
“It hasn’t come into the public eye as much as I would have expected it to, given how important it is.”
“Understanding how we can reverse changes in viscoelasticity will literally provide us with a multitude of therapeutic avenues for reducing the effects of age-related diseases, of which there are a truly many.
“I think it should be a really important focus for science going forward.”

Professor Justin Cooper-White
Head of School
School of Chemical Engineering
Faculty of Engineering, Architecture and Information Technology
Affiliate Professor
Australian Institute of Bioengineering and Nanotechnology
Words: Harriet Dempsey-Jones
Image Credits: Anjanette Hudson


